 The same applies to lead compounds such as lead acetate, lead oxide, lead nitrate, and lead carbonate. Lead salts are attributed to water hazard class 2, and consequently are harmful. Lead(II)salts and organic lead compounds are most harmful ecotoxicologically. Lead and lead compounds are generally toxic pollutants. What are the environmental effects of lead in water? In The Netherlands, lead in food causes about half of the human lead exposure. Generally, lead dissolved or suspended in wastewater mostly stems from streets, pipes and soils. 
In architecture lead is applied in roofs and in stained glass windows. Fortunately, this form of release of lead is less and less abundant. This organic lead compounds is quickly converted to inorganic lead, and ends up in water, sometimes even in drinking water. The lead compound tetra-ethyl lead is applied as an additive in fuels. Most industrially processed lead is applied for fabricating computer and TV screens. Organic lead is applied in petroleum production, and inorganic lead compounds are applied for battery and paint production. Selling tubes of lead white is now prohibited in the European Union. It is no longer applied because of its extreme toxicity. Lead white, an alkalic lead carbonate (2PbCO 3. In Rome lead was often released as a by-product of silver mining. Water pollution containing lead compounds derived from lead ores in the mining industry was first mentioned by architect Vitruvius, in 20 B.C., when he gave out a warning of its health effects. The Romans often filled the pipes with wine on holidays, causing the layer to dissolve and form lead sugar.ĭespite its toxicity, the above-mentioned lead(II)acetate was applied far into the 19th century, to sweeten wine and other beverages and food stuffs. This layer functions as a protective coating for the underlying lead of the pipes. Inside the pipes, a layer of hardly soluble alkalic lead carbonate is formed. Lead may bind to carbonate, therefore lower amounts of lead dissolve in hard water. Lead from pipes may partially dissolve in the water flowing through. 
Lead waterworks were often applied in former days, and these may still be present in old buildings. Lead compounds are generally soluble in soft, slightly acidic water. In these forms lead is extremely insoluble, and is present as immobile compounds in the environment. Lead frequently binds to sulphur in sulphide form (S 2-), or to phosphor in phosphate form (PO 4 3-). A well-known example of a water soluble lead compound is lead sugar (lead(II)acetate), which derived its name from its sweet nature. It may however occur dissolved in water as PbCO 3 or Pb(CO 3) 2 2. When both oxygen and water are present, metallic lead is converted to lead hydroxide (Pb(OH) 2):ĢPb(s)+ O 2(g) + 2H 2O(l) -> 2 Pb(OH) 2(s)Įlementary lead does not dissolve in water under normal conditions (20 oC, and pressure = 1 bar). A small lead oxide (PbO) layer forms at the surface of the metal. 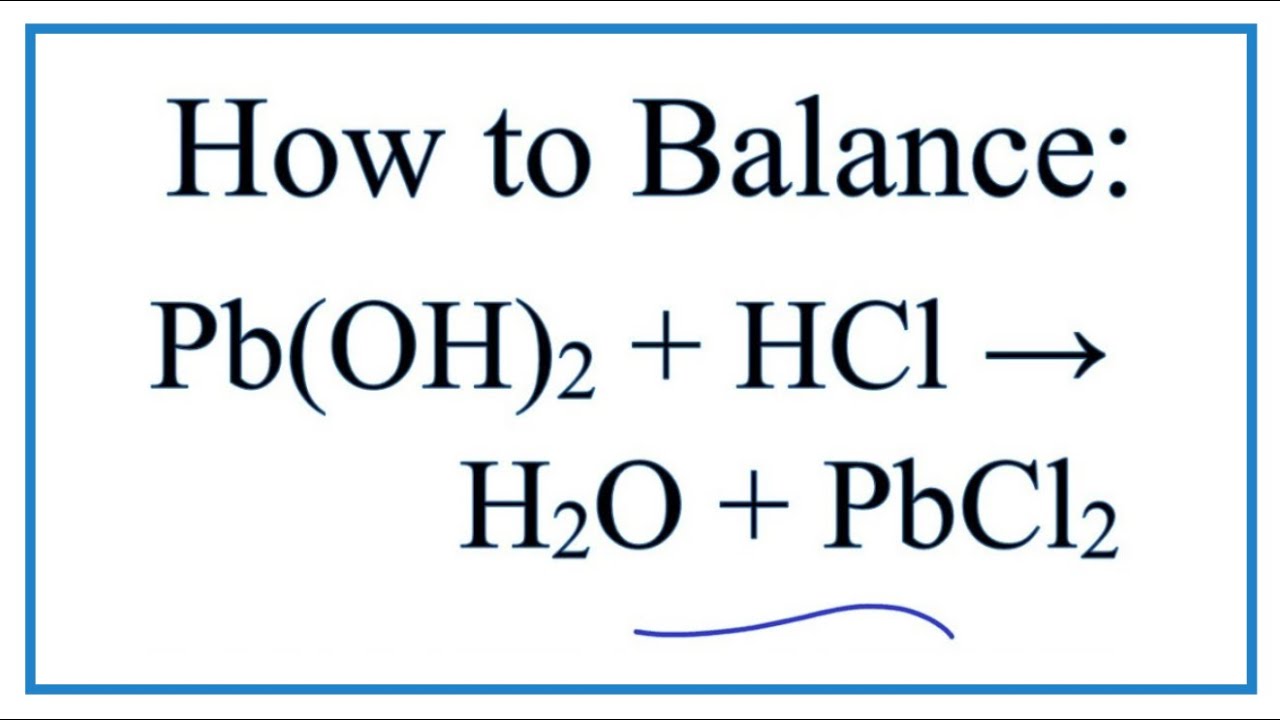
However, when lead comes in contact with moist air reactivity with water increases. Under normal conditions lead does not react with water. In what way and in what form does lead react with water? 
The World Health Organization (WHO) stated a legal limit of 50 ppb for lead in 1995, which is decreased to 10 ppb in 2010. Phytoplankton contains approximately 5-10 ppm lead (dry mass), freshwater fish approximately 0.5-1000 ppb, and oyster approximately 500 ppb. On average rivers contain between 3 and 30 ppb. Lead (Pb) and water Lead and water: reaction mechanisms, environmental impact and health effects Seawater contains trace amounts of lead (2-30 ppt).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
